Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Sasaki, Yuji; Ban, Yasutoshi; Morita, Keisuke; Matsumiya, Masahiko*; Ono, Ryoma*; Shiroishi, Hidenobu*
Solvent Extraction Research and Development, Japan, 27(1), p.63 - 67, 2020/00
Times Cited Count:6 Percentile:31.74(Chemistry, Multidisciplinary)Mutual separation technique of Dy and Nd in Nd magnet is studied. Dy is more valuable than Nd, then Dy might be isolated and reused. Lanthanide elements can be extracted thoroughly by diglycolamide (DGA) extractants, we use this reagent for the recovery and isolation of Dy. Tetradodecyl-DGA (TDdDGA) has relatively high separation factors(SF) between Dy and Nd (SF=17-18) in HNO extraction system, counter-current extraction using TDdDGA was applied for their mutual separation. From the present study, using the condition, four extraction stages, organic phase: 0.1M TDdDGA in n-dodecane, aqueous phase: 0.3M HNO
extraction system, counter-current extraction using TDdDGA was applied for their mutual separation. From the present study, using the condition, four extraction stages, organic phase: 0.1M TDdDGA in n-dodecane, aqueous phase: 0.3M HNO , 92% Dy can be recovered with 0.7% co-extraction of Nd.
, 92% Dy can be recovered with 0.7% co-extraction of Nd.
 -isopropylacrylamide) with diglycolamide-typed ligands
-isopropylacrylamide) with diglycolamide-typed ligandsSaga, Kaname*; Suzuki, Hideya; Matsumura, Tatsuro; Tsukahara, Takehiko*
Analytical Sciences, 35(4), p.461 - 464, 2019/04
Times Cited Count:3 Percentile:7.76(Chemistry, Analytical)The phase transition-based gelification phenomenon of poly- -isopropylacrylamide (PNIPAAm) in aqueous solutions has a great potential in developing new waste-free extraction processes of metal ions. By using hydrophobic diglycolamide-typed ligands in gelification extraction, a one-step complete extraction of all the RE ions from a nitric acid solution was successfully realized.
-isopropylacrylamide (PNIPAAm) in aqueous solutions has a great potential in developing new waste-free extraction processes of metal ions. By using hydrophobic diglycolamide-typed ligands in gelification extraction, a one-step complete extraction of all the RE ions from a nitric acid solution was successfully realized.
Kaneko, Masashi; Watanabe, Masayuki; Matsumura, Tatsuro
Dalton Transactions, 45(43), p.17530 - 17537, 2016/11
Times Cited Count:33 Percentile:87.63(Chemistry, Inorganic & Nuclear)Relativistic density functional calculations were applied to study the separation behaviors of Am(III) ion from Eu(III) ion by diglycolamide (DGA) and nitrilotriacetamide (NTA) ligands in order to understand the difference in the separation mechanism of their reagents. The complexation reaction was modeled on the basis of previous experimental studies. The calculated energies based on stabilization by complex formation at the ZORA-B2PLYP/SARC level predicted that the DGA reagent preferably coordinated to Eu(III) ion when compared with Am(III) ion. In contrast, the NTA reagent selectively coordinated to Am(III) ion when compared with Eu(III) ion. These results reproduced the experimental selectivity of DGA and NTA ligands toward Eu(III) and Am(III) ions. Mulliken's population analyses implied that the difference in the contribution of the bonding property between the f-orbital of Am and donor atoms determined the comparative stability of Eu and Am complexes.
Sasaki, Yuji; Sugo, Yumi; Morita, Keisuke; Nash, K.*; Nash, K. L.*
Solvent Extraction and Ion Exchange, 33(7), p.625 - 641, 2015/10
Times Cited Count:95 Percentile:91.19(Chemistry, Multidisciplinary)The effect of alkyl substituents at amidic N atoms in diglycolamide (DGA) compounds on solvent extraction was investigated. The solubility in water and n-dodecane, loading capacity, and distribution ratio (D) of lanthanides and actinides for various DGA compounds were monitored. DGA derivatives with short alkyl chains, for example methyl and ethyl groups, are highly water soluble, while DGA derivatives with long alkyl chains are highly soluble in n-dodecane. The loading capacity correlats with their alkyl chain lengths. The results of masking effect and solubility tests indicate that TEDGA(tetraethyl-diglycolamide) is the best actinide masking agent among the water-soluble DGA derivatives. Actinide extractions were performed using ten DGA compounds in six diluents (nitrobenzene, 1,2-dichloroethane, 1-octanol, chloroform, toluene, and n-dodecane), in which it was observed that DGA derivatives with shorter alkyl chains show higher D values.
Tachimori, Shoichi; Suzuki, Shinichi; Sasaki, Yuji; Apichaibukol, A.
Solvent Extraction and Ion Exchange, 21(5), p.707 - 715, 2003/09
Times Cited Count:48 Percentile:75.63(Chemistry, Multidisciplinary)no abstracts in English
 and UO
and UO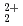 ions with tridentate ligand diglycolamide (DGA)
ions with tridentate ligand diglycolamide (DGA)Hirata, Masaru; Guilbard, P.*; Dobler, M.; Tachimori, Shoichi
Physical Chemistry Chemical Physics, 5(4), p.691 - 695, 2003/02
Times Cited Count:20 Percentile:54.61(Chemistry, Physical)no abstracts in English
Sugo, Yumi; Sasaki, Yuji; Tachimori, Shoichi
Radiochimica Acta, 90(3), p.161 - 165, 2002/04
Times Cited Count:153 Percentile:99.24(Chemistry, Inorganic & Nuclear)no abstracts in English
Sasaki, Yuji; Tachimori, Shoichi
Solvent Extraction and Ion Exchange, 20(1), p.21 - 34, 2002/01
Times Cited Count:186 Percentile:95.52(Chemistry, Multidisciplinary)no abstracts in English
 -n-dodecane system
-n-dodecane systemSasaki, Yuji; Sugo, Yumi; Suzuki, Shinichi; Tachimori, Shoichi
Solvent Extraction and Ion Exchange, 19(1), p.91 - 103, 2001/01
Times Cited Count:626 Percentile:99.62(Chemistry, Multidisciplinary)no abstracts in English
Sasaki, Yuji; Sugo, Yumi; Tachimori, Shoichi
Proceedings of International Conference on Scientific Research on the Back-end of the Fuel Cycle for the 21st Century (ATALANTE 2000) (Internet), 6 Pages, 2000/10
no abstracts in English
Yaita, Tsuyoshi
Kinzoku Sakutai No Bunshi Sekkei Wakushoppu -(II) Hobunshu, p.63 - 75, 2000/06
no abstracts in English
Yaita, Tsuyoshi
Proceedings of International Youth Nuclear Congress 2000 (CD-ROM), 4 Pages, 2000/00
no abstracts in English
Sasaki, Yuji; Tsubata, Yasuhiro; Morita, Keisuke; Sugo, Yumi; Suzuki, Tomoya
no journal, ,
no abstracts in English
Morita, Keisuke; Sasaki, Yuji; Suzuki, Tomoya
no journal, ,
no abstracts in English
Sasaki, Yuji; Sugo, Yumi; Morita, Keisuke
no journal, ,
The effect of the alkyl substituents on amidic N atoms in diglycolamide (DGA) compounds on solvent extraction has been investigated. The purpose of this study is to examine the effect of alkyl substituents on Ln and An extraction. Diglycolamides obtained are O(CH CONR
CONR )
) with R = CH
with R = CH , C
, C H
H , C
, C H
H , C
, C H
H , C
, C H
H , C
, C H
H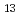 , C
, C H
H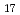 , C
, C H
H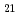 , C
, C H
H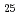 and alkyl and phenyl-groups. From the present work, it was clear that DGA with shorter alkyl chain have high solubility in water, and those with longer alkyl chain have high solubility in n-dodecane. Although DGA with short alkyl chain have high distribution ratio, DGA with long alkyl chain have high loading capacity of Nd. Even DGA derivatives, DGA with phenyl groups have not only low solubility in dodecane but also low D values for lanthanides.
and alkyl and phenyl-groups. From the present work, it was clear that DGA with shorter alkyl chain have high solubility in water, and those with longer alkyl chain have high solubility in n-dodecane. Although DGA with short alkyl chain have high distribution ratio, DGA with long alkyl chain have high loading capacity of Nd. Even DGA derivatives, DGA with phenyl groups have not only low solubility in dodecane but also low D values for lanthanides.
Sasaki, Yuji; Ban, Yasutoshi; Morita, Keisuke; Matsumiya, Masahiko*; Ono, Ryoma*; Shiroishi, Hidenobu*
no journal, ,
Mutual separation technique of Dy and Nd in Nd magnet is noticed for the recycle of ranthanide elements. Both Dy and Nd belong to ranthanide elements, then the mutual separation is very hard. Lanthanide elements can be extracted thoroughly by diglycolamide (DGA) extractants, we use this reagent for the recovery and isolation of Dy. Tetradodecyl-DGA (TDdDGA) has relatively high separation factors (SF) between light and heavy ranthanides, this compound is useful for this purpose. In this work, the counter-current extraction using TDdDGA was applied for their separation, and the results will be discussed.
Sasaki, Yuji; Ban, Yasutoshi; Morita, Keisuke; Matsumiya, Masahiko*
no journal, ,
Although rare earth elements show low concentration in the crust, these metals are available widely in the industrial field. It is important to know the better methods for mutual separation and recovery of the rare earth elements. These elements in nitric acid can be extracted by diglycolamide compounds, however, it shows low mutual separation ratio. Then in order to increase the separation efficiency, multi-step counter-current extraction is applied. In this presentation, the basic results concerning this extraction method will be discussed.
Sasaki, Yuji; Matsumiya, Masahiko*
no journal, ,
DGA (diglycolamide) compounds can extract lanthanide (Ln) elements in nitric acid and shows the high D(Ln). The separation factors (SF) between light and heavy Ln are relatively high, thus the mutual separation of Ln can be easily done by DGA compounds. On the other hand, their SF is not so high, the separation and recovery between Nd and Dy are performed using batchwise multi-step extraction. In this work, calculation results are compared with the experiments, and its effect is confirmed.
Sasaki, Yuji; Morita, Keisuke; Matsumiya, Masahiko*
no journal, ,
The separation and isolation of Dy from Nd magnet is important task for the rare-metal recycles. These lanthanide metal ions have normally low separation factors because of same oxidation state and similar ionic radii. Therefore, multi-step extractions are indispensable in order to increase mutual separation factors. We performed the batchwise multi-step extraction for the mutual separation of Nd and Dy using diglycolamide extractant. The analytical results will be shown in the poster presentation.
Sasaki, Yuji; Matsumiya, Masahiko*; Nakase, Masahiko*
no journal, ,
We will show the background how to prepare DGA compounds that we develop initially and the results of their extraction abilities. In addition, as an example of utility of DGA compounds for the mutual separation of lanthanides and actinides, the results of the batchwise multi-step extraction for Nd and Dy separation will be shown.